Nrf2 and the Keap1/Nrf2, Nrf2/sMaf/ARE Pathways
Bridging the gap between normal redox homeostasis and oxidative stress lies a transcription factor protein known as nuclear factor erythroid 2 (Nrf2).
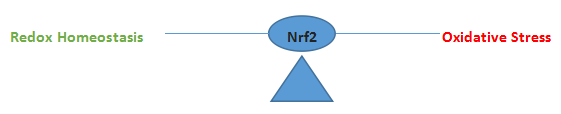
Nrf2 is a basic leucine zipper (bZIP) protein functioning to regulate the expression of many antioxidant proteins. Nrf2 exists in the cytosol where it is bound to the KEAP1 regulatory molecule. In this condition Nrf2 cannot cross into the nucleus. Under conditions of oxidative stress however, KEAP1 releases its hold on Nrf2 allowing the protein it to translocate into the nucleus where it binds to small Maf proteins and the resulting Nrf2/sMaf heterodimer in turn binds to the Antioxidant Response Element (ARE) on various stress related gene targets. In recent years Nrf2/KEAP1 regulation of the Nrf2/sMAF/ARE transcription pathway has been investigated extensively to assess its possible role in aging and disease. Many studies are showing a positive correlation between Nrf2 nuclear translocation and amelioration of disease resulting in increased interest in Nrf2 promotors and KEAP1 agonists as possible therapeutic agents. Other studies show paradoxical outcomes when considering Nrf2 in relation to cancer. In that case, tumor cells are able to use the Nrf2 system as a means to upregulate antioxidant expression which appears to confer additional chemo-protective properties possibly making the tumor more virulent and resistant to therapy.
Left unchecked, elevated levels of pro-oxidants are capable of affecting changes in lipids, proteins and
DNA, which can be toxic and lead to disease and/or environmental decline. For this reason, oxidative
stress has historically been studied mostly for its negative effects and indeed it has been shown to be
involved with heart, liver, lung and neurologic diseases among others. More recently however, it has
been discovered that brief and controlled levels of oxidative stress are a fundamental component of
many cell signaling processes including many involved in regulating expression and function of key
proteins. This relatively new finding is taking the study of oxidative stress in new directions, wherein
researchers believe that oxidative regulation of protein function and the genetic mechanisms of
oxidative stress response may hold the keys to ameliorating the effects of aging and disease.
Among the key stress response elements known to be regulated by Nrf2 include:





